Model of solidification and crystal growth
Useful thermodynamic relations for phase change
Specific heat (dim \([\text{E}]/([\text{M}].[\Theta])\)
Latent heat at melting temperature \(T_m\) (dim [E]/[M])
Thermodynamic Maxwell relations
Proof of Eq. (581) with \(\mathcal{U}(V,S)\)
Proof of Eq. (583) with \(\mathcal{F}(V,T)\)
Solidification of a pure substance and Stefan’s problem
We consider at the melting temperature \(T_m\), the coexistence of a chemical specie \(A\) into two phases: a tiny solid seed inside a liquid bath of same specie. The temperature of the system \(T\) is quenched below the melting temperature \(T<T_m\). Then the solidification occurs: the small seed grows i.e. the liquid becomes solid and the excess of latent heat \(\mathcal{L}\) is released at the interface and advected with the normal velocity of the interface \(v_n\). The modeling of that phase change problem is performed with the classical Stefan’s problem.
Stefan’s problem of phase change
The Stefan’s model of phase change is composed of three equations: the heat equation with two conditions at interface between solid and liquid:
where \(T\) is the temperature, \(\Lambda_{\Phi}\) is the thermal conductivity for both phases \(\Phi=0,1\). The first interface condition is the Stefan condition (conservation at interface):
where \(\hat{\boldsymbol{n}}\) is the normal vector at interface, and \(v_{n}\) the normal velocity of the interface, \(\mathcal{L}\) is the latent heat. The second interface condition is the Gibbs-Thomson condition:
where \(T_i\) is the temperature at the interface, \(T_{m}\) is the melting temperature, \(\sigma\) is the surface tension, \(\mathcal{L}\) is the latent heat, \(\kappa\) is the curvature, \(m_{k}\) is the interface mobility, and \(v_{n}\) is the normal velocity of interface.
Derivation of phase-field model
The derivation of the phase-field model of solidification is inspired from [1]. For derivation of crystal growth model see [2] and [3].
Free energy density \(\mathscr{F}\)
The Stefan’s problem is a “sharp interface” modelling. Here, an equivalent diffuse interface is derived. We introduce a phase-field \(\psi(\boldsymbol{x},t)\) with \(\psi=-1\) in the solid phase and \(\psi=+1\) in the liquid phase. The solid (respectively liquid) is characterized by its specific heat \(C_p^s\) (resp. \(C_p^l\)) and thermal conductivity \(\Lambda^s\) (resp. \(\Lambda^s\)). The interface is characterized by its normal velocity \(v_n\), the unit normal vector \(\boldsymbol{n}_{\psi}\) and the latent heat \(\mathcal{L}\). The solidification problem is presented on Fig. Fig. 63
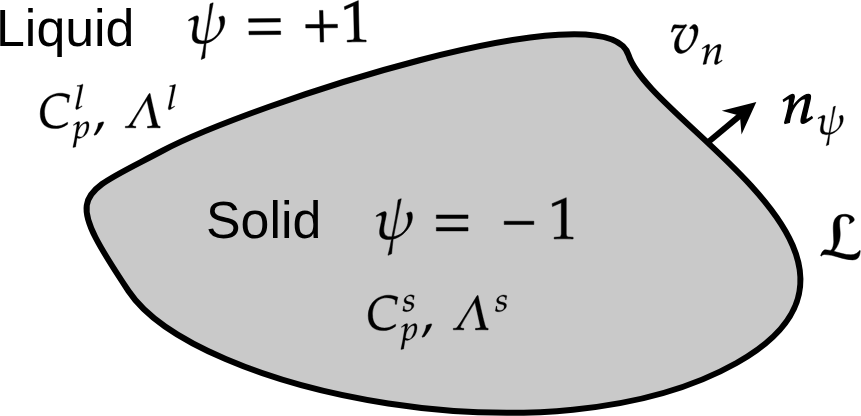
Fig. 63 Solidification
Functional of free energy
Functional of free energy
The free energy density is now the functional of two functions \(\psi\) and \(T\)
(588)\[\mathscr{F}[\psi,{\color{red}T}]=\int\Bigl[\underbrace{\mathcal{F}_{int}(\psi,\boldsymbol{\nabla}\psi)}_{\text{Standard interface free energy}}+\underbrace{{\color{red}f_{bulk}(\psi,T)}}_{\text{Thermodynamic of bulks}}\Bigr]dV\]That free energy density contains two contributions: the first one is the interface contribution \(\mathcal{F}_{int}(\psi,\boldsymbol{\nabla}\psi)\) and the second one the bulk free energy density \(f_{bulk}(\psi,T)\).
Interface contribution
The first one is standard and has been widely discussed until now. It is defined by a double-well plus a gradient energy term:
\[\mathcal{F}_{int}(\psi,\boldsymbol{\nabla}\psi)=f_{dw}(\psi)+\frac{\zeta}{2}(\boldsymbol{\nabla}\psi)^{2}\]where the double-well is defined by
\[\begin{split}f_{dw}(\psi)&=Hg_{3}(\psi)\\ &=H(\psi-\psi^{\star})^{2}(\psi+\psi^{\star})^{2}\end{split}\]the minima are \(\psi^{\star}=\pm1\). With that double-well free energy, the Euler-Lagrange solution and the interface have already been discussed in Alternative forms of double-wells: the equilibrium solution is \(\psi^{eq}(x)=\psi^{\star}\tanh\left(2x/W\right)\), the interface width is \(W=\frac{1}{\psi^{\star}}\sqrt{\frac{2\zeta}{H}}\) and the surface tension is \(\sigma=\frac{4}{3}\sqrt{2\zeta H}\).
Bulk contribution
The bulk free energy density takes into account the free energy of each phase \({\color{red}f_{s}(T)}\) in the solid and \({\color{red}f_{l}(T)}\) in the liquid:
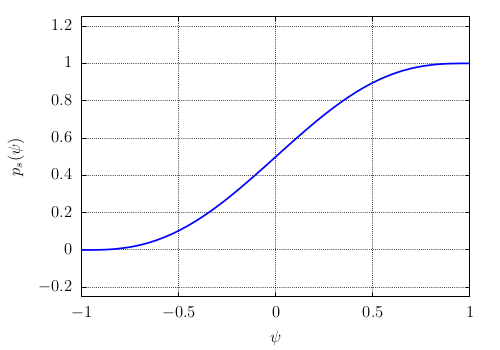
(589)\[f_{bulk}(\psi,T)=p_{s}(\psi){\color{red}f_{s}(T)}+\left[1-p_{s}(\psi)\right]{\color{red}f_{l}(T)}\]where \(p_{s}(\psi)\) is an interpolation polynomial defined by:
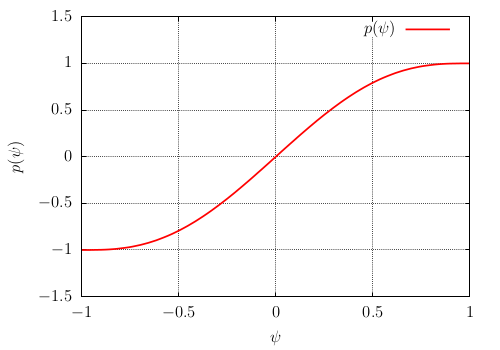
(590)\[p_{s}(\psi)=\frac{1+p(\psi)}{2}\]of minimum and maximum values between 0 and 1 (see Fig. Fig. 64). It is defined by \(p(\psi)\) of minimum and maximum values -1 and +1 (see Fig. Fig. 65)
(591)\[p(\psi)=\frac{15}{8}\left[\psi-\frac{2}{3}\psi^{3}+\frac{\psi^{5}}{5}\right]\]
Variation of \(\mathscr{F}\)
The variation of \(\mathscr{F}\) writes:
Temperature equation
Temperature equation
The conservation of internal energy density \(e\) writes:
where the Fourier law has been assumed for the diffusive flux. After expressing the internal energy \(e(\phi,s)\) as a function of \(T\) (see proof below), we obtain:
Proof
Variation of \(\mathscr{F}\) wrt \(T\): opposite of entropy
(595)\[\begin{split}s(\psi,T)&=-\frac{\delta\mathscr{F}}{\delta T}\\ &=-\frac{\partial f_{bulk}(\psi,T)}{\partial T}\\ &=-p_{s}(\psi)\underbrace{\frac{\partial f_{s}}{\partial T}}_{s_{s}(T)}-\left[1-p_{s}(\psi)\right]\underbrace{\frac{\partial f_{l}}{\partial T}}_{s_{l}(T)}\end{split}\]Thermo identity (\(de=Tds\)) and chain rule
\[\begin{split}de&=Tds(\psi,T)\\ &=\underbrace{T\frac{\partial s}{\partial T}}_{C_{p}\text{: specific heat}}dT+T\underbrace{\frac{\partial s}{\partial\psi}}_{\text{use previous Eq.}}d\psi\end{split}\]For the first term of the right-hand side we use the definition of specific heat Eq. (). For the second term we derive Eq. () wrt \(\psi\). After division by \(dt\), we obtain
(596)\[\frac{\partial e}{\partial t} =C_{p}\frac{\partial T}{\partial t}+p_{s}^{\prime}(\psi)\underbrace{T\left[s_{l}(T)-s_{s}(T)\right]}_{\equiv\mathcal{L}\text{: latent heat}}\frac{\partial\psi}{\partial t}\]
Phase-field equation
The dynamic of \(\psi\) must obey to the following Allen-Cahn equation Eq. (452) to minimize the free energy:
To calculate the variation of \(\mathscr{F}\) wrt \(\psi\), we use Eq. ():
To simplify the thermodynamic driving source, a common hypothesis is to consider the temperature \(T\) close to the melting temperature \(T_m\). A Taylor expansion yields:
where \(\Phi=s,l\). At \(T_m\), \(f_l(T_m)=f_s(T_m)\) and the latent heat is () \(\mathcal{L}=T_{m}[s_{l}(T_{m})-s_{s}(T_{m})]\). Finally
In the right-hand side, the coefficient \(H\) is set in factor:
and we define new coefficients:
Finally, using Eqs. (600), (601) and (602) in Eq. (599) we obtain for the phase-field equation:
Summary
Phase-field model of solidification
The phase-field model of solidification is composed of one phase-field equation for the interface:
coupled with one temperature equation:
Equivalence between phase-field model and Stefan’s problem
Equivalence between sharp and diffuse interface models
The sharp interface model (Stefan’s problem) and the phase-field model expressed with the dimensionless temperature \(\theta=(T-T_{m})C_{p}/\mathcal{L}\) write:
where \(\theta\) is the dimensionless temperature, \(d_{0}=\sigma_{0}T_{m}C_{p}/\mathcal{L}^{2}\) is the capillary length, and \(\beta=C_{p}/\mathcal{L}m\) is the kinetic coefficient. With the matched asymptotic expansions, it is proved that both models are equivalent provided that the two following conditions hold:
where \({\color{teal}a_{1}}=0.8839\) and \({\color{teal}a_{2}}=0.6267\) (see [3]). In the right-hand side of Eqs. () and () \(\lambda\), \(W_{0}\) and \(\tau_{0}\) are the parameters of the phase-field model.
Anisotropy function and phase-field model of crystal growth
Dependence of properties with normal vector \(\boldsymbol{n}\equiv\boldsymbol{n}_{\psi}=-\boldsymbol{\nabla}\psi/\bigl|\boldsymbol{\nabla}\psi\bigr|\)
where the anisotropy function \(a_{s}(\boldsymbol{n})\) depends on the shape of crystal we want to simulate. Two anisotropy functions are presented below for main growing directions [100] and [110].
where \(\epsilon_s\) and \(\delta\) are positive parameters.
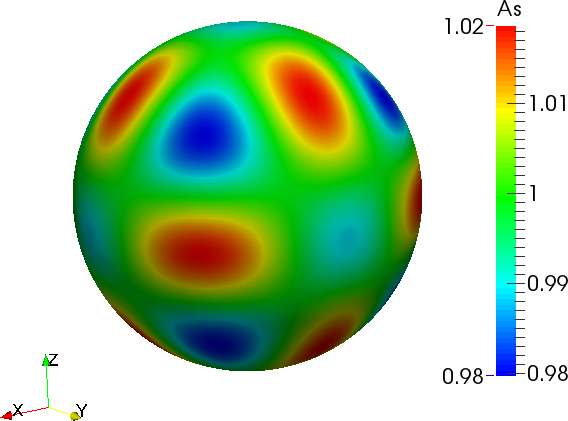
Phase-field model of crystal growth
Phase-field equation
where
and the anisotropy function \(a_s(\boldsymbol{n})\) is defined by Eq. (611).
Temperature equation
The temperature equation remains unchanged:
References
Section author: Alain Cartalade