Mass transfer through interfaces
Gibbs-Thomson condition
Gibbs-Thomson condition for a curved interface
Gibbs-Thomson condition
For a curved interface at equilibrium:
where the index \(i\) is for “interface”. The pressure difference is given by the Laplace’s law:
With its definition:
With its def (Eq. ([eq:Def_p-c_local])), Eq. ([eq:Equilib_p_Curved]) writes
Using common tangent construction Eq. (387) for \(f_{dw}(\phi_{out})-f_{dw}(\phi_{in})\):
and finally:
This is the Gibbs-Thomson condition which means that the equilibrium chemical potential of a curved interface is equal to the equilibrium chemical potential for a flat interface corrected by the curvature and the surface tension.
Ostwald ripening
Consequence of Gibbs-Thomson condition with \(\boldsymbol{j}_{CH}\): Ostwald ripening
Ostwald ripening
The Gibbs-Thomson condition writes:
If we consider two droplets of radius \(R_{1}\) and \(R_{2}\) with \(R_{1}<R_{2}\), Eq. () implies:
There exists a flux from \(\mu_{i}(R_{1})\) to \(\mu_{i}(R_{2})\) because \(\boldsymbol{j}_{CH}\propto-\boldsymbol{\nabla}\mu_{\phi}(\boldsymbol{x},t)\) (see Fig. Fig-Ostawald-Ripening). Consequence: the smallest droplet disappears and the biggest one grows. This is the Ostwald ripening.
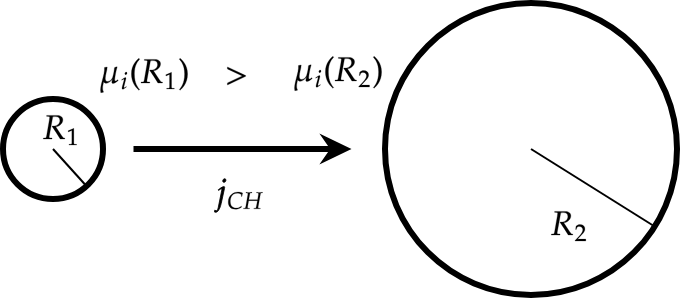
Fig. 62 Principle of Ostwald ripening